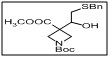
1-(tert-butyl) 3-methyl 3-(2-(benzylthio)-1-hydroxyethyl)azetidine-1,3-dicarboxylate
The benzylthio group acts as a protected thiol, while the hydroxyethyl moiety provides a reactive site for further functionalization, including etherification, esterification, or coupling reactions.

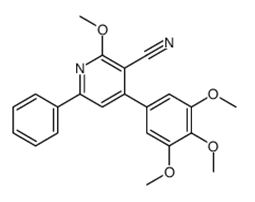
![Methyl 2-[6-(2-chlorophenyl)-4-(4-chlorophenyl)-3-cyanopyridin-2-yl]sulfanylacetate](https://navyachempharma.com/wp-content/uploads/2025/08/18-8.png)
Reviews
There are no reviews yet.