Products
- Catalog
Leading Specialty Chemical Solutions
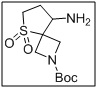
tert-butyl 8-amino-5-thia-2-azaspiro[3.4]octane-2-carboxylate 5,5-dioxide
CAS No:
- 1340481-83-5
Category:
- The tert-butyl ester protects the carboxyl functionality while leaving the amino group free for functionalization, enabling amide bond formation, coupling reactions, or derivatization.
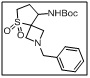
tert-butyl (2-benzyl-5,5-dioxido-5-thia-2-azaspiro[3.4]octan-8-yl)carbamate
CAS No:
Category:
- The Boc group protects the amino functionality, while the benzyl and sulfone substitutions provide reactivity for further derivatization, enabling the synthesis of bioactive spirocyclic scaffolds with structural rigidity and stereochemical integrity.
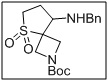
tert-butyl 8-(benzylamino)-5-thia-2-azaspiro[3.4]octane-2-carboxylate 5,5-dioxide
CAS No:
Category:
- The sulfone and spirocyclic core offer structural rigidity and bioactive potential for downstream applications.
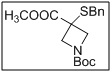
1-(tert-butyl) 3-methyl 3-(benzylthio)azetidine-1,3-dicarboxylate
CAS No:
Category:
- The benzylthio group provides a protected thiol functionality for downstream transformations, while the dual ester protection allows selective deprotection and functionalization.
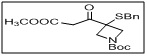
tert-butyl 3-(benzylthio)-3-(3-methoxy-3-oxopropanoyl)azetidine-1-carboxylate
CAS No:
Category:
- The benzylthio group allows selective functionalization, and the acyl ester functionality provides reactivity for further transformations, including nucleophilic substitutions or coupling reactions.
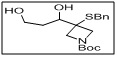
tert-butyl 3-(benzylthio)-3-(1,3-dihydroxypropyl)azetidine-1-carboxylate
CAS No:
Category:
- The benzylthio group provides a protected thiol functionality, while the dihydroxypropyl moiety offers reactive sites for selective functionalization or derivatization.
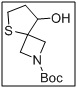
tert-butyl 8-hydroxy-5-thia-2-azaspiro[3.4]octane-2-carboxylate
CAS No:
Category:
- The spirocyclic core offers structural rigidity and stereochemical control, making it ideal for downstream pharmaceutical applications.
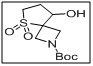
tert-butyl 8-hydroxy-5-thia-2-azaspiro[3.4]octane-2-carboxylate 5,5-dioxide
CAS No:
Category:
- The sulfone and spirocyclic core impart structural rigidity, stability, and bioactive potential, making it suitable for pharmaceutical and synthetic applications.
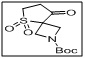
tert-butyl 8-oxo-5-thia-2-azaspiro[3.4]octane-2-carboxylate 5,5-dioxide
CAS No:
Category:
- This compound is a valuable intermediate in medicinal chemistry, peptide synthesis, and spirocyclic drug development.
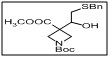
1-(tert-butyl) 3-methyl 3-(2-(benzylthio)-1-hydroxyethyl)azetidine-1,3-dicarboxylate
CAS No:
Category:
- The benzylthio group acts as a protected thiol, while the hydroxyethyl moiety provides a reactive site for further functionalization, including etherification, esterification, or coupling reactions.
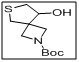
tert-butyl 8-hydroxy-6-thia-2-azaspiro[3.4]octane-2-carboxylate
CAS No:
Category:
- The tert-butyl ester protects the carboxyl functionality, while the hydroxy group provides a reactive site for functionalization, including etherification, esterification, or coupling reactions.
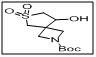
tert-butyl 8-hydroxy-6-thia-2-azaspiro[3.4]octane-2-carboxylate 6,6-dioxide
CAS No:
Category:
- The sulfone group and spirocyclic structure impart stability, rigidity, and stereochemical control, making it ideal for synthesizing bioactive molecules and pharmaceutical intermediates.
