Products
- Catalog
Leading Specialty Chemical Solutions
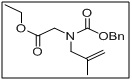
ethyl N-((benzyloxy)carbonyl)-N-(2-methylallyl)glycinate
CAS No:
Category:
- This compound serves as a versatile intermediate in peptide synthesis, medicinal chemistry, and chiral molecule synthesis.
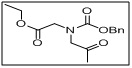
ethyl N-((benzyloxy)carbonyl)-N-(2-oxopropyl)glycinate
CAS No:
Category:
- This compound is a versatile intermediate in peptide synthesis, medicinal chemistry, and chiral molecule synthesis.
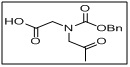
N-((benzyloxy)carbonyl)-N-(2-oxopropyl)glycine
CAS No:
Category:
- The carboxylic acid functionality provides a versatile site for amide bond formation and other derivatizations.
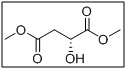
dimethyl (R)-2-hydroxysuccinate
CAS No:
- 70681-41-3
Category:
- Its chiral hydroxyl functionality allows for enantioselective transformations, making it valuable for synthesis of bioactive molecules, chiral ligands, and intermediates for active pharmaceutical ingredients (APIs).
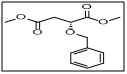
dimethyl (R)-2-(benzyloxy)succinate
CAS No:
- 134356-95-9
Category:
- The benzyloxy group acts as a protecting group for the hydroxyl functionality, enabling selective transformations, while the R-chirality provides stereochemical control in downstream reactions.
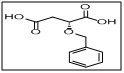
(R)-2-(benzyloxy)succinic acid
CAS No:
Category:
- This compound is a valuable intermediate in asymmetric synthesis, medicinal chemistry, and fine chemical production.
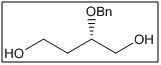
(S)-2-(benzyloxy)butane-1,4-diol
CAS No:
Category:
- The benzyloxy group provides protection for the secondary alcohol, enabling selective transformations, while the S-chirality allows stereochemical control in downstream reactions.
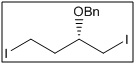
(S)-(((1,4-diiodobutan-2-yl)oxy)methyl)benzene
CAS No:
Category:
- The diiodo moiety allows for selective cross-coupling reactions (e.g., Suzuki, Sonogashira, Heck) and other halogen-based transformations.
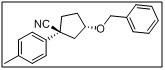
(1S,3S)-3-(benzyloxy)-1-(p-tolyl)cyclopentane-1-carbonitrile
CAS No:
Category:
- The benzyloxy group acts as a protecting group for the secondary alcohol, while the nitrile functionality is suitable for further transformations such as hydrolysis, amidation, or cyclization.
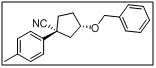
(1S,3S)-3-(benzyloxy)-1-phenylcyclopentane-1-carbonitrile
CAS No:
Category:
- The benzyloxy group acts as a protecting group for the secondary alcohol, while the nitrile functionality allows for further functionalization, including hydrolysis, amidation, or cyclization.
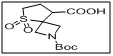
2-(tert-butoxycarbonyl)-5-thia-2-azaspiro[3.4]octane-8-carboxylic acid 5,5-dioxide
CAS No:
Category:
- This compound is a valuable intermediate in medicinal chemistry, peptide synthesis, and spirocyclic compound development.

tert-butyl 8-(((benzyloxy)carbonyl)amino)-5-thia-2-azaspiro[3.4]octane-2-carboxylate 5,5-dioxide
CAS No:
Category:
- This compound is a highly versatile intermediate in peptide synthesis, medicinal chemistry, and spirocyclic drug development.
